INTRODUCTION: Myelodysplastic/myeloproliferative neoplasm with ring sideroblasts and thrombocytosis (MDS/MPN-RS-T) is a rare hematological disorder characterized by anemia, bone marrow dysplasia with ring sideroblasts and persistent thrombocytosis, and high frequency of SF3B1 and JAK2 mutations. Despite clinical, histological and molecular similarities with MDS with ring sideroblasts (MDS-RS), the clinical outcomes of these entities are diverse. To date, there is no data evaluating specific functional pathways which might explain phenotypic and clinical differences beyond diverse frequencies of JAK2 mutation.
METHODS: We evaluated a total of 24 patients (pts) with MDS/MPN-RS-T and 27 pts with MDS-RS. Diagnosis was based on WHO 2017 criteria and confirmed by two independent hematopathologists. Whole bone marrow DNA was subject to 81 gene targeted next-generation sequencing (NGS) analysis. CD34+ cells from bone marrow samples of 4 pts with MDS/MPN-RS-T, 7 pts with MDS-RS and 17 healthy individuals obtained from AllCells (Emeryville, CA) were isolated using the CD34 MicroBead Kit and RNA was isolated using the PicoPure RNA isolation kit. Fastq files were mapped to the human genome (build GRCh38) in TopHat2 using the default options. Differential gene expression analysis was conducted using DESeq2 in R version 3.6.2. Pathway enrichment analysis was performed using gene set enrichment analysis, with the fgsea library in R.
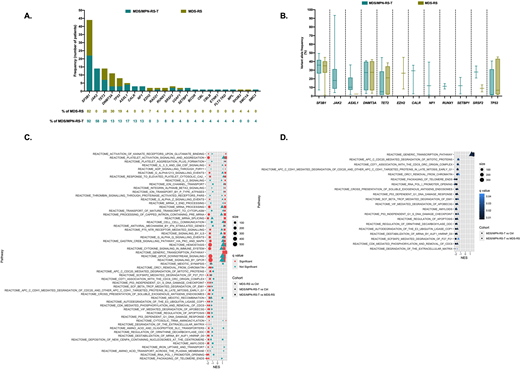
RESULTS: Patients with MDS/MPN-RS-T had higher median bone marrow ring sideroblast percentage (47% vs 32%, p=0.04) and absolute neutrophil count (4.34x109/L vs 2.99x109/L, p=0.001). Frequency of identified mutations and their VAFs compared to MDS-RS are shown in Figure 1A. The median number of mutations was higher in MDS/MPN-RS-T than in MDS-RS (3 vs 2, p<0.001). SF3B1 mutations were the most frequent in both entities (MDS/MPN-RS-T: 92%, MDS-RS: 82%), had similar median VAF (34% vs 32%, p=0.619), and involved the hot spot codon K700E in 64% and 43% of MDS-RS and MDS/MPN-RS-T (p=0.227), respectively. As expected, 58% of pts with MDS/MPN-RS-T had JAK2 V617F mutations but were also more likely to have mutations in kinase signaling genes (NF1, SETBP1, CBL, CBLB, FLT3 TKD, MPL) compared to MDS-RS (29% vs 4%, p=0.019). Four (40%) of JAK2 negative MDS/MPN-RS-T had mutations in kinase signaling genes. There were no differences in frequency of TET2 mutations between both entities. However, there was a trend for the median VAF of TET2 mutations in MDS/MPN-RS-T to be lower than in MDS-RS (1.5% vs 21.1%, p=0.177) suggesting a likely subclonal nature of these mutations compared to MDS-RS in which they appeared as dominant events. MDS/MPN-RS-T showed distinct transcriptomic profile compared to both healthy controls and MDS-RS. Compared to healthy controls, a total of 2 pathways were significantly upregulated and 58 were downregulated in MDS/MPN-RS-T while 5 pathways were upregulated and 69 were downregulated in MDS-RS. Compared to MDS-RS, a total of 29 pathways were significantly upregulated and 26 were downregulated in MDS/MPN-RS-T. The most significantly upregulated pathways in MDS/MPN-RS-T included genes involved in platelet activation and aggregation, cytokine signaling, and signaling through GPC receptors (Figure 1C). Compared to both healthy control and MDS-RS, MDS/MPN-RS-T was characterized by downregulation of genes involved in DNA damage response, regulation of apoptosis, telomere maintenance and RNA synthesis (Figure 1D). MDS-RS was characterized by downregulation of genes involved in signaling by GPC receptors and MAPK signaling, mRNA splicing, cytokine signaling and signaling through interleukins compared to both control and MDS/MPN-RS-T (Figure 1C).
CONCLUSIONS: MDS/MPN-RS-T is characterized by co-dominance of SF3B1 and JAK2 mutations and presence of minor kinase signaling mutations not observed in MDS-RS. Upregulation of cytokine and interleukin signaling mediated through GPC receptors, and downregulation of genes involved in apoptosis and DNA damage are unique transcriptomic features of MDS/MPN-RS-T likely driven by genotype. These unique genomic and transcriptomic characteristics of MDS/MPN-RS-T supports the classification of MDS/MPN-RS-T based on genomic features beyond presence of SF3B1 mutation, and might represent potential therapeutic avenues for this rare disease.
Sasaki:Otsuka: Honoraria; Pfizer Japan: Consultancy; Novartis: Consultancy, Research Funding; Daiichi Sankyo: Consultancy. Kantarjian:Sanofi: Research Funding; Abbvie: Honoraria, Research Funding; Janssen: Honoraria; BMS: Research Funding; Actinium: Honoraria, Membership on an entity's Board of Directors or advisory committees; Adaptive biotechnologies: Honoraria; Aptitute Health: Honoraria; Immunogen: Research Funding; Jazz: Research Funding; Daiichi-Sankyo: Honoraria, Research Funding; Amgen: Honoraria, Research Funding; BioAscend: Honoraria; Novartis: Honoraria, Research Funding; Delta Fly: Honoraria; Pfizer: Honoraria, Research Funding; Oxford Biomedical: Honoraria; Ascentage: Research Funding. Garcia-Manero:Astex Pharmaceuticals: Consultancy, Honoraria, Research Funding; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Amphivena Therapeutics: Research Funding; Acceleron Pharmaceuticals: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Research Funding; Onconova: Research Funding; Merck: Research Funding; Novartis: Research Funding; H3 Biomedicine: Research Funding; Helsinn Therapeutics: Consultancy, Honoraria, Research Funding; Jazz Pharmaceuticals: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal